Wells at Greatest Risk of Contamination from Wildfires Include:
- Dug, sandpoint, or any wells that do not have a watertight casing.
- Wells that do not have a watertight, secure cover or cap.
- Wells constructed prior to 1986. *Prior to water well construction regulations in Nebraska, construction practices did not have mandatory grout seal in the annular space around the well casing, making it less resistant to having contaminants or surface runoff from firefighting efforts to enter the well.
Quick facts…
- Fire damage to ground level and underground structures can lead to water contamination in a well and malfunction of both water well and wastewater systems. If damage is found, contact your Nebraska licensed water well contractor and/or Nebraska certified onsite wastewater contractor.
- Owners need to inspect and test well and wastewater systems following wildfire impact.
- Keep bottled water on hand for cooking and drinking until well water has been tested and determined safe to use.
- Wildfire impacts to well water quality used for irrigation will most likely be from high levels of sediment and ash, and a potential change in pH. Changes in pH can temporarily affect the availability for some nutrients for plants. If the water already has a high sodium absorption ratio, it can increase the sodium damage as well. The high concentrations of ash and sediment is often more of an issue with the physical irrigation system itself.
- If ponds or water tanks have been adversely affected by post-fire ash, fire retardant – both aerial and foam, and/or sediment, provide an alternative water source for livestock until steps have been taken to assure water quality. Water tanks will need to be thoroughly scrubbed and cleaned.
- Remember that most water contaminants are colorless, odorless, and tasteless therefore testing water quality is of utmost importance.
If you are the owner of a private domestic water well, livestock well, or irrigation well that was in the path of a wildfire, there are legitimate concerns about fire-related impacts to your well, water, and even wastewater systems. Nebraska Extension recommends well owners perform a visual inspection of their wellhead, water and wastewater system using the Nebraska Extension Rapid Assessment Form for Wells & Onsite Wastewater Systems Affected by Wildfire. If you find damage to any of the following, you should contact a Nebraska licensed water well contractor and/or a Nebraska certified onsite wastewater contractor to do a more in depth assessment of the damage and make repairs.
- The wellhead, secure sanitary cap, screen, electrical conduit.
- The electrical wires and connectors that supply power to your well.
- The well casing and any above-ground piping that brings water to your home, buildings, or fields (in the case of irrigation wells).
- The well house or irrigation pump and equipment.
- The equipment such as water treatment equipment and electronic controls.
- The pressure tanks potentially caused by exposure to excessive heat.
- The vents, storage tanks, and/or overflow pipes.
- Heat damage to plastic pipes PEX, HDPE, PP, PVC, and CPVC could leach VOCs into your drinking water.
- The septic tank covers, drop boxes, inspection ports, and or component access areas. This includes both heat damage to components, as well ash and debris that could have entered the system causing a blockage.
- All septic tanks should be thoroughly pumped out.
- The above and below ground onsite system components. Wet spots, sewage odors, or hearing odd sounds from mechanical components can be indicators of structural issues.
- The piping that enters the home/building.
- The area of the drainfield and septic tank from debris or being run over by heavy equipment. Make sure good grass/vegetation cover is in place to protect from erosion.
- Make sure that wastewater is moving throughout the system as it should.
Water Taste and Odors
Once you know that water lines are undamaged, you may still notice that your water looks or smells earthy, smoky, or burnt. The water may be visually cloudy, have sediment in it, or be noticeably discolored, such as a reddish tint due to the iron oxide in aerial fire retardants. Fire retardants are made up of ammonium-based compounds that contain phosphate or sulfate. It is highly recommended to thoroughly flush your water lines and divert the flush water away from your onsite wastewater system. Due to the compounds often found in fire retardants, if it was used within the vicinity of a wellhead, it is advised to monitor for ammonia and nitrate for several months after a wildfire event. Testing or heavy metals should also be considered as they can also be components of fire retardants. If a sewage smell is detected this could be an indicator of a structural issue which a Nebraska certified onsite wastewater professional will need to help assess.
Inside Your Home
To the extent you can, visually check the water-supply system, including plumbing, for any damage, signs of leaks, or changes in operation. You should check to see if your well and plumbing system maintained positive pressure during the fire. This can be done by turning on a faucet in the household and observing if water flows. You should not hear any air being released from the faucet such as a sputtering sound, nor should you hear water hammering, a loud hammering/knocking/banging noise from the pipes. The water should flow steadily and uninterrupted. If you do hear a sputtering sound from the faucet with water intermittently spurting out when it is turned on, and/or a water hammering sound, this is an indication that your well and household plumbing had a loss of pressure and may have been damaged.
If You Have Had Loss of Water Pressure
If you have had a loss of pressure or the water system has been damaged, it is likely that your water may be contaminated with bacteria. Damaged well components should be repaired or replaced by a Nebraska licensed water well contractor. Anytime a water system loses pressure, the water should be tested for the presence of bacteria before it is used for drinking or cooking. Anytime the well or water system is repaired, it should be disinfected after a repair is made and then tested to ensure the water is safe for drinking or cooking.
Testing Your Well Water for Bacteria, Nitrate, and Other Contaminants of Concern
Water quality knowledge for owners of a private domestic water well, livestock well, or irrigation well is a critical component in best protecting the health of one’s family, livestock, and environment. We have known, naturally occurring contaminates in our groundwater and surface water throughout the state, as well as contaminants introduced by human activity. When a disastrous event like a wildfire occurs, other contaminants pose legitimate concerns about fire related impacts to one’s well, water resources, and wastewater systems. Nebraska Extension recommends well owners test their private well water quality for the following:
Contaminant & Parameters | Wildfire Related Source |
| Total Dissolved Solids and Total Suspended Solids (TDS and TSS) | Ash resulting from burned biomass |
| Bacteria (total coliform, E. coli) preferably by the Colilert Quantitray method to get a number result | Infiltration after depressurization of water systems from power loss |
| Nitrate, nitrite, ammonia (NO3- , NO2- , NH3) | Aerial fire retardants |
Volatile Organic Compounds (VOCs) *If there is known heat damage to PVC well casing and or secure cap seals. | Melted PVC from well casings |
Per- and Polyfluoroalkyl Substances (PFAS) *If there is know use of aqueous firefighting foam used in the vicinity of the wellhead. | Fire suppression chemicals, Aqueous firefighting foams |
| Metals - Arsenic (As), Chromium (Cr), Lead (Pb), Magnesium (Mg) | Combustion of biomass and other burned infrastructure |
Water Testing Kits
Drinking water from private wells is just as important as water from public water systems. For this reason, Nebraska Extension highly recommends that drinking water samples from private wells be tested at a certified laboratory. The State of Nebraska Public Health Environmental Laboratory (NPPHEL) certifies other public health environmental laboratories in Nebraska for drinking water analysis of public water systems throughout the state.
When testing drinking water for the presence of bacteria, nitrate, or other contaminants of concern, it is necessary to collect samples in the appropriate container(s) provided by the laboratory. Carefully follow the instructions that come with the kit(s) when collecting a sample, label the bottles accordingly, fill out the testing forms completely, and return the full kit(s) to the laboratory in the time window outlined within the instructions. See the table below of the certified laboratories that accept private well samples.
When ordering a bacteriological kit from any of the laboratories listed below, request a Bacteria Kit that will give a numerical count result, otherwise known as a Coliform by Quantitray test.
Certified Public Health Environmental Laboratories in Nebraska that Accept Private Well Samples |
Name of Lab | Phone # | Address | Certified for Analytes |
| Central District Health Department | 308-385-5175 | 1137 South Locust Grand Island, NE 68801 | Total Coliform/E.coli by Colilert SM9223B |
| Enviro Services Inc. | 308-632-3933 | 818 S. Beltline Hwy. East Scottsbluff, NE 69361 | Total Coliform/E.coli by Colilert SM9223B |
| Midwest Laboratories, Inc. (by reciprocity) | 402-334-7770 | 13611 B Street Omaha, NE 68144 | Total Coliform/E.coli by Quanti-tray 2000SM9223B, Nitrate as N by EPA 300.0,Nitrite as N by SM4500-NO2-B, Nitrate/Nitrite as N by EPA 353.2, Copper by EPA 200.8, Lead by EPA 200.7, VOCs by EPA 524.2, Fluoride by EPA 300.0 |
| American Agricultural Laboratory | 308-345-3670 | 700 East D St. McCook, NE 69001 | Total Coliform/E.coli by Colilert SM9223B Nitrate/Nitrite EPA 353.2 |
| Nebraska Public Health Environmental Lab | 402-471-3935 | 3701 South 14th St. Lincoln, NE 68502 | All (unless noted by lab) – Nebraska’s primary state-certified accrediting laboratory |
Using Your Water While You Wait for Test Results
You can use your water for short showers and flushing toilets for solid waste, not each time if it is liquid waste only. Take care to avoid swallowing water from showers or baths. Limit laundry washing. You should not use your water for drinking or cooking purposes unless you have received test results back that confirm it is safe to consume. This caution includes not washing dishes or other cooking utensils in it as well.
Disinfecting Your Water Well and Distribution System
After conducting the Rapid Assessment of your water well, and confirming that the electrical components of your well can be safely operated, you can begin preparing to disinfect your well by shocking it. Check with the manufacturer of water softeners, sand filters, iron removal filters, adsorptive media such as activated carbon filters, reverse osmosis filters, or any other water treatment equipment in place prior to shock chlorinating the system. Water softeners, sand filters, and iron removal filters can often be backwashed with a strong chlorine solution. Adsorptive media such as activated carbon, and reverse osmosis filters should generally be removed from the distribution system during shock chlorination and until the chlorine has been flushed from the distribution system.
Follow the shock chlorination steps below.
| Table I. Volume of water contained per foot of well depth. | |
Well casing diameter | Water volume per foot of |
4 | 0.65 |
| 1Volume of water calculated as the volume of a cylinder in cubic feet multiplied by 7.48 gallons/cubic foot. | |
Step 1. Determine the diameter of the well. Measure the inside diameter of the well in inches.
Step 2. Determine the volume of water per foot for the well. Using Table I, find the volume of water per foot for your well. Use the diameter of the well determined in Step 1. Find the gallons per foot for that well diameter. Example: The well is measured to have a 6-inch diameter. The gallons per foot of depth for a 6-inch well is 1.47 gallons.
For large diameter wells or cisterns, contact the Nebraska Department of Water, Environment, and Energy for information on how to disinfect your system.
Step 3. Determine the depth of water in the well. The company that constructed the well should be able to provide you with the well depth and water level. Example: The well is 50 feet deep and the water level is at 20 feet. The well contains 30 feet of water (50-20=30 feet)
Step 4. Determine the total gallons of water in the well. Multiply the depth of water in the well determined in Step 3 by the gallons of water per foot for your well determined in Step 2. This is the total gallons of water in the well. Example: Multiply 30 feet (the depth of the water in the well) by 1.47 gallons of water per foot to get 44 gallons of well water (30 x 1.47 = 44 gallons of water in the well)
Step 5. Estimate the volume of water in the distribution system. Total the water storage in the system, including the water heater, pressure tank, etc., and add 50 gallons for the pipeline. Example: The system has a 30-gallon hot water heater and a 30-gallon pressure tank. 30 gallons (water heater) + 30 gallons (pressure tank) + 50 gallons (pipeline) = 110 gallons in the distribution system.
Step 6. Determine the water contained in the entire system. Add the water volume in the well determined in Step 4 and the water contained in the distribution system determined in Step 5 to determine the total water volume to be shock chlorinated. Example: 44 gallons in the well + 110 gallons in the distribution system = 154 total gallons to be shock chlorinated
| ||||||||||||||||
Step 7. Determine the amount of chlorine product required for a 200 ppm solution. Table II lists the product amounts needed to create a 200 ppm free chlorine solution in each 100 gallons of water using typically available sources. Determine the amount of product necessary for shock chlorination of your system by dividing the total water volume by 100; then multiplying this number by the amount of product needed per 100 gallons. Use the worksheet at the end of this guide. Example: For our example we will use liquid household bleach containing 5 percent to 6 percent sodium hypochlorite (NaOCl); Divide 154 (total gallons) by 100 = 1.54 100-gallon units. Multiply 1.54 100-gallon units times 3 pints/100-gallon units = 4.62 pints. You would need about 4 1/2 pints of liquid household bleach containing 5 percent to 6 percent NaOCl.
If you use liquid chlorine bleach, select a product that contains 5 percent to 6 percent NaOCl. Check the product label or call the manufacturer’s toll free number provided on the label to confirm the NaOCl concentration of a particular product. While many household bleaches contain 5 percent to 6 percent NaOCl, household bleach products are also available with considerably lower concentrations, including products with 2.5 percent to 3 percent NaOCl. Avoid bleaches with fragrances, soaps, or other additives. Use fresh, newly purchased bleach since bleach loses its effectiveness with age and with exposure to light or heat.
When chlorine is added to water it reacts with certain components in the water including iron, manganese, sulfur, microorganisms, plant material, and others. These components “tie up” some of the chlorine, making it unavailable for disinfection. The chlorine that does not react with these components is available for disinfection. This is called free chlorine. The amounts recommended in Table II should provide sufficient free chlorine for most drinking water supplies, including those containing minerals. Concerned individuals can test the chlorine concentration by using test kits available from some plumbing, home improvement, and water supply equipment dealers. Be certain the kit tests free chlorine, not total chlorine.
The disinfecting effectiveness of chlorine is impacted by water temperature and pH. A well professional may treat for these parameters when shock chlorinating a well. A homeowner will most likely not be able to treat for these water quality parameters when performing shock chlorination, and the amounts recommended in Table II should provide sufficient free chlorine in most situations.
Step 8: Introduce the chlorine material into the well and distribution system. The best way to introduce chlorine material into the well is to dissolve the predetermined amount of chlorine in a 5-gallon bucket of fresh water. Be sure the bucket is plastic and has been thoroughly washed. Pour the chlorine solution into the well. Try to splash the solution on the sidewalls of the well casing as much as possible. Attach a hose to the water hydrant or faucet nearest the well and run water through the hydrant and back into the well (Figure 1). This will thoroughly mix the chlorine solution and well water.
If using solid forms of chlorine, dissolve tablets or powder/granular in a bucket of water and introduce into the well casing as described for using liquid chlorine sources. Again, use a nearby hydrant and hose to circulate water through a portion of the distribution system to assure that the chlorine material is thoroughly mixed with well water.
Regardless of how you introduce the chlorine material into your well, start and stop the pump several times to ensure that the chlorine is thoroughly mixed with well water. Recirculate the water until a strong chlorine smell has been noted for at least five minutes.
After the chlorine has been placed in the well and the casing, etc., has been washed down, move around the water distribution system and open each faucet (hot and cold), hydrant or other water outlet. Allow water to flow until a strong chlorine odor reaches that position in the system. Then close the valve at that location. Do this with all faucets, hydrants and other outlets in the system.
If a strong chlorine odor is not detected at each site, add more chlorine to the well. This may be an indication that the product has lost effectiveness or is not of the concentration it was thought to be.
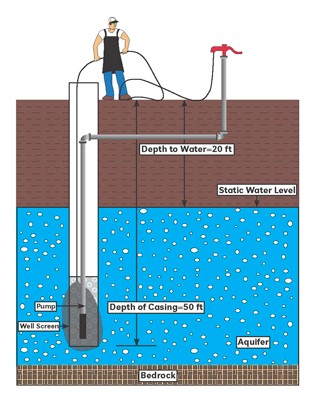
Figure 1. Recirculating water through a nearby hydrant after the introduction of concentrated chlorine into a well in order nto thoroughly mix chlorine with the well water.
Step 9: Let the chlorine disinfect the system. The disinfecting effectiveness of chlorine depends on the amount of time the available chlorine is in contact with the water and plumbing (contact time). To allow adequate disinfection contact time, refrain from using water from the well for at least two to three hours, preferably overnight. Longer contact times will result in a more effective disinfection of the system.
Step 10: Flush the system to remove the chlorine. After the water system chlorination has been completed, the entire system must be emptied of chlorine and thoroughly flushed with fresh water. Run water out of each faucet or hydrant until the chlorine odor dissipates. Distribute the wastewater on gravel roads or other areas without plants or aquatic life, which it might harm.
| If your house is served by a septic system for wastewater treatment, do not allow more than 50 gallons of chlorinated water to enter the septic system. If possible, attach a hose to outlets inside the house and distribute the water to a non-grass area away from the house. The chlorine will eventually evaporate. |
Step11: Retest the water supply for bacterial contamination. The final step is to retest the water to ensure that the water source is coliform, fecal, and/or E. coli bacteria free. Take a water sample one to two weeks after shock chlorinating the well, using the same procedures as before. Though most shock chlorination treatments are successful, do not drink the water until the laboratory results confirm that no coliform, fecal, or E. coli bacteria are present. Retest the well every month for two to three months to be sure contamination is not reoccurring. If test results are negative, an annual water analysis program can be reinstated.
If the water supply continues to develop coliform, fecal, or E. coli bacterial contamination problems after being shock chlorinated, other options must be considered. Options might include repairing the well, or constructing a new well. Continuous disinfection of the water supply using continuous chlorination, distillation, ozonation, or ultraviolet radiation may be an option. See Drinking Water Treatment: An Overview (EC703), Drinking Water Treatment: Distillation (G1493) and Drinking Water Treatment: Continuous Chlorination (G1496) for more information on these continuous disinfection options.
Nuisance iron, manganese, and sulfur bacteria are more resistant to shock chlorination than other types of bacteria. You may have to repeat the shock chlorination procedure several times to manage these bacteria. In addition, these types of bacteria tend to recur, so periodic chlorination may be needed to control the problem.
Chlorine Solution Calculation Worksheet
| Process | Example: | Your well: | |
| 1. | Diameter of well: | 6 inches | __________inches |
| 2. | Volume of water per foot: (See Table I) | 1.47 gallons | __________gallons |
| 3a. | Depth of casing: | 50 feet | __________feet |
| 3b. | Depth to water: | 20 feet | __________feet |
| 3c. | Total depth of water: (3a - 3b) | 30 feet | __________feet |
| 4. | Total volume of water in casing: (No. 2 x No. 3c) | 44 gallons | __________gallons |
| 5. | Volume of water in the system: | 110 gallons | __________gallons |
| 6. | Total volume of water: (No. 4 + No. 5) | 154 gallons | __________gallons |
Calculate Amount of Chlorine Product for a 200 ppm Free Chlorine Solution: | |||
Chlorine product used: liquid household bleach | |||
| 7a. | Product needed per 100 gallons: | 3 pints | __________(ounces/pints) |
| 7b. | Total product needed: (No. 6 divided by 100) x No. 7a | 4.5 pints | __________(ounces/pints) |
To best protect the health of your family if you have wildfire damage to your well and/or onsite wastewater system, Nebraska Extension strongly recommends contacting your local Nebraska licensed well or certified wastewater professional to further assess the system(s) and make necessary repairs.